Abstract
Research Article
Comparison of resting-state functional and effective connectivity between default mode network and memory encoding related areas
Supat Saetia*, Fernando Rosas, Yousuke Ogata, Natsue Yoshimura and Yasuharu Koike
Published: 24 April, 2020 | Volume 4 - Issue 1 | Pages: 029-037
Currently brain connectivity modelling, constructed from data acquired by non-invasive technique such as functional magnetic resonance imaging (fMRI), is a well-received approach to illustrate brain function. However, not all connectivity models contains equal amount of information. There are two types of connectivity model that could be constructed from fMRI data, functional and effective connectivity. Effective connectivity includes information about the direction of the connection, while functional connectivity does not. This makes interpretation of effective connectivity more meaningful than functional connectivity. The objective of this study is to show the improvement in interpretability of effective connectivity model in comparison to functional connectivity model. In this study, we show how the difference in the information contained within these two model impacts the interpretation of the resulting connectivity model by analyzing resting-state fMRI data on episodic memory-related cognitive function using CONN Toolbox bivariate correlation measurement for functional connectivity analysis and Tigramite causal discovery framework for effective connectivity analysis on an episodic memory related resting-state fMRI dataset. The comparison between functional and effective connectivity results show that effective connectivity contains more information than the functional connectivity, and the difference in the information contained within these two types of model could significantly impact the intepretation of true brain function. In conclusion, we show that for the connectivity between specific pair of brain regions, effective connectivity analysis reveals more informative characteristic of the connectivity in comparison to functional connectivity where the depicted connectivity lack any additional characteristic information such as the direction of the connection or whether it is a unidirectional or bidirectional. These additional information improve interpretability of brain connectivity study. Thus, we would like to emphasis the important of brain function study using effective connectivity modelling to obtain valid interpretation of true brain function as currently a large body of research in this field focuses only on functional connectivity model.
Read Full Article HTML DOI: 10.29328/journal.jnnd.1001031 Cite this Article Read Full Article PDF
References
- Tulving E. What Is Episodic Memory? Current Directions in Psychological Science. 1993; 2: 67-70.
- Ullman MT. Contributions of memory circuits to language: the declarative/procedural model, Cognition. 2004; 92: 231-270. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15037131
- Rubinov M, Sporns O. Complex network measures of brain connectivity: uses and interpretations. NeuroImage. 2010; 52, 1059-1069. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/19819337
- Friston KJ. Functional and effective connectivity in neuroimaging: A synthesis. Human Brain Mapping. 1994; 2: 56-78.
- Granger CWJ. Investigating Causal Relations by Econometric Models and Cross-spectral Methods. Econometrica. 1996; 37: 424.
- Pearl J. Causality: Models, Reasoning and Inference (Cambridge University Press, New York, NY, USA). 2009.
- Whitfield-Gabrieli S, Nieto-Castanon A. Conn: a functional connectivity toolbox for correlated and anticorrelated brain networks. Brain connectivity. 2012; 2: 125-141. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/22642651
- Runge J. Quantifying information transfer and mediation along causal pathways in complex systems. Phys Rev E. 2015; 92: 062829. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26764766
- Runge J. Causal network reconstruction from time series: From theoretical assumptions to practical estimation. Chaos. 2018; 28: 075310. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/30070533
- Margulies DS, Bottger J, Long X, Lv Y, Kelly C, et al. Resting developments: a review of fMRI post-processing methodologies for spontaneous brain activity. Magma. 2010; 23: 289-307. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/20972883
- Duggento A, Passamonti L, Valenza G, Barbieri R, Guerrisi M, et al. Multivariate Granger causality unveils directed parietal to prefrontal cortex connectivity during task-free MRI. Sci Rep. 2018; 8: 5571. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/29615790
- Corkin S, What's new with the amnesic patient H.M.? Nat Rev Neurosci. 2002; 3: 153-160. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/11836523
- Henson RN, Rugg MD, Shallice T, Dolan RJ. Confidence in recognition memory for words: dissociating right prefrontal roles in episodic retrieval. J Cogn Neurosci. 2000; 12: 913-923. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/11177413
- Runge J, Nowack P, Kretschmer M, Flaxman S, Sejdinovic D. Detecting causal associations in large nonlinear time series datasets. Sci Adv. 2019; 27; 5. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/31807692
- Fox MD, Raichle M. Spontaneous fluctuations in brain activity observed with functional magnetic resonance imaging. Nat Rev Neurosci. 2007; 8: 700-711. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/17704812
- Behzadi Y, Restom K, Liau J, Liu TT. A component based noise correction method (CompCor) for BOLD and perfusion based fMRI. NeuroImage. 2007; 37: 90-101. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/17560126
- Laufs H, Krakow K, Sterzer P, Eger E, Beyerle A, et al. Electroencephalographic Signatures of Attentional and Cognitive Default Modes in Spontaneous Brain Activity Fluctuations at Rest. Proc Natl Acad Sci U S A. 2003; 100: 11053-11058. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/12958209
- Fox M, Snyder AZ, Vincent JL, Corbetta M, Van Essen DC, et al. The Human Brain Is Intrinsically Organized into Dynamic, Anticorrelated Functional Networks. Proc Natl Acad Sci U S A. 2005; 102: 9673-9678. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15976020
- Greicius MD, Krasnow B, Reiss AL, Menon V. Functional Connectivity in the Resting Brain: A Network Analysis of the Default Mode Hypothesis. Proc Natl Acad Sci U S A. 2003; 100: 253-258. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/12506194
- Fransson P. Spontaneous low-frequency BOLD signal fluctuations: An fMRI investigation of the resting-state default mode of brain function hypothesis. Hum Brain Mapp. 2005; 26: 15-29. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15852468
- Vincent JL, Snyder AZ, Fox MD, Shannon BJ, Andrews JR. Coherent Spontaneous Activity Identifies a Hippocampal-Parietal Memory Network. J Neurophysio. 2006; 96: 3517-3531. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/16899645
- Rombouts SARB, Stam CJ, Kuijer JPA, Scheltens P, Barkhof F. Identifying confounds to increase specificity during a "no task condition". NeuroImage. 2003; 20: 1236-1245. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/14568492
- Horwitz B. The elusive concept of brain connectivity. NeuroImage. 2003; 19: 466-470.
- O'Kane G, Kensinger EA, Corkin S. Evidence for semantic learning in profound amnesia: An investigation with patient H.M. Hippocampus. 2004; 14; 417-425. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15224979
- Lehn H, Steffenach HA, van Strien NM, Veltman DJ, Witter MP. A specific role of the human hippocampus in recall of temporal sequences. The J Neurosci. 2009; 29: 3475-3484. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/19295153
- Merkow MB, Burke JF, Kahana MJ. The human hippocampus contributes to both the recollection and familiarity components of recognition memory. Proc Natl Acad Sci U S A. 2015; 112: 14378-14383. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26578784
- Harand C, Bertran F, La Joie R, Landeau B. The Hippocampus Remains Activated over the Long Term for the Retrieval of Truly Episodic Memories. PloS one. 2012; 7: e43495). PubMed: https://www.ncbi.nlm.nih.gov/pubmed/22937055
- Ezzati A, Katz MJ, Zammit AR, Lipton ML. Zimmerman ME, et al. Differential association of left and right hippocampal volumes with verbal episodic and spatial memory in older adults. Neuropsychologia. 2016; 93: 380-385. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/27542320
- Van Hoesen GW. The parahippocampal gyrus: New observations regarding its cortical connections in the monkey. Trends in Neurosciences. 1982; 5: 345-350.
- Bohbot VD, Allen JJB, Dagher A, Dumoulin SO, Evans AC, et al. Role of the parahippocampal cortex in memory for the configuration but not the identity of objects: converging evidence from patients with selective thermal lesions and fMRI. Frontiers in human neuroscience. 2015; 9: 431. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26283949
- Kohler S, Danckert S, Gati JS, Menon RS. Novelty responses to relational and non-relational information in the hippocampus and the parahippocampal region: A comparison based on event-related fMRI. Hippocampus. 2005; 15: 763-774. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15999342
- WIttmann BC, Bunzeck N, Dolan RJ, Du E. Anticipation of novelty recruits reward system and hippocampus while promoting recollection. NeuroImage. 2007; 38: 194-202. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/17764976
- Howard LR, Kumaran D, Olafsdottir HF, Spiers HJ. Double Dissociation between Hippocampal and Parahippocampal Responses to Object-Background Context and Scene Novelty. J Neurosci. 2011; 31: 5253-5261. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/21471360
- Nielsen FA, Balslev D, Hansen LK. Mining the posterior cingulate: Segregation between memory and pain components. NeuroImage. 2005; 27: 520-532. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15946864
- Sestieri C, Corbetta M, Romani GL, Shulman GL. Episodic Memory Retrieval, Parietal Cortex, and the Default Mode Network: Functional and Topographic Analyses. J Neurosci. 2011; 31: 4407-4420. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/21430142
- Raichle ME, MacLeod AM, Snyder AZ, Powers WJ, Gusnard DA, et al. A default mode of brain function. Proceedings of the National Academy of Sciences. 2001; 98: 676-682.
- Buckner RL, Andrews-Hanna JR, Schacter DL. The brain's default network. Annals of the New York Academy of Sciences. 1124; 1-38.
- Gusnard DA, Akbudak E, Shulman GL, Raichle ME. Medial prefrontal cortex and self-referential mental activity: relation to a default mode of brain function. Proc Natl Acad Sci U S A. 2001; 98: 4259-4264. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/11259662
- Addis DR, Wong AT, Schacter DL. Remembering the past and imagining the future: Common and distinct neural substrates during event construction and elaboration. Neuropsychologia. 2007; 45: 1363-1377. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/17126370
- Leech R, Kamourieh S, Beckmann CF, Sharp DJ. Fractionating the Default Mode Network: Distinct Contributions of the Ventral and Dorsal Posterior Cingulate Cortex to Cognitive Control. J Neurosci. 2011; 31: 3217-3224. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/21368033
- Mason MF, Norton MI, Van Horn JD, Wegner DM, Grafton ST, et al. Wandering Minds: The Default Network and Stimulus-Independent Thought. Science. 2007; 315: 393-395. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/17234951
- Tulving E. What Is Episodic Memory? Current Directions in Psychological Science. 1993; 2: 67-70.
- Ullman MT. Contributions of memory circuits to language: the declarative/procedural model, Cognition. 2004; 92: 231-270. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15037131
- Rubinov M, Sporns O. Complex network measures of brain connectivity: uses and interpretations. NeuroImage. 2010; 52, 1059-1069. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/19819337
- Friston KJ. Functional and effective connectivity in neuroimaging: A synthesis. Human Brain Mapping. 1994; 2: 56-78.
- Granger CWJ. Investigating Causal Relations by Econometric Models and Cross-spectral Methods. Econometrica. 1996; 37: 424.
- Pearl J. Causality: Models, Reasoning and Inference (Cambridge University Press, New York, NY, USA). 2009.
- Whitfield-Gabrieli S, Nieto-Castanon A. Conn: a functional connectivity toolbox for correlated and anticorrelated brain networks. Brain connectivity. 2012; 2: 125-141. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/22642651
- Runge J. Quantifying information transfer and mediation along causal pathways in complex systems. Phys Rev E. 2015; 92: 062829. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26764766
- Runge J. Causal network reconstruction from time series: From theoretical assumptions to practical estimation. Chaos. 2018; 28: 075310. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/30070533
- Margulies DS, Bottger J, Long X, Lv Y, Kelly C, et al. Resting developments: a review of fMRI post-processing methodologies for spontaneous brain activity. Magma. 2010; 23: 289-307. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/20972883
- Duggento A, Passamonti L, Valenza G, Barbieri R, Guerrisi M, et al. Multivariate Granger causality unveils directed parietal to prefrontal cortex connectivity during task-free MRI. Sci Rep. 2018; 8: 5571. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/29615790
- Corkin S, What's new with the amnesic patient H.M.? Nat Rev Neurosci. 2002; 3: 153-160. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/11836523
- Henson RN, Rugg MD, Shallice T, Dolan RJ. Confidence in recognition memory for words: dissociating right prefrontal roles in episodic retrieval. J Cogn Neurosci. 2000; 12: 913-923. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/11177413
- Runge J, Nowack P, Kretschmer M, Flaxman S, Sejdinovic D. Detecting causal associations in large nonlinear time series datasets. Sci Adv. 2019; 27; 5. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/31807692
- Fox MD, Raichle M. Spontaneous fluctuations in brain activity observed with functional magnetic resonance imaging. Nat Rev Neurosci. 2007; 8: 700-711. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/17704812
- Behzadi Y, Restom K, Liau J, Liu TT. A component based noise correction method (CompCor) for BOLD and perfusion based fMRI. NeuroImage. 2007; 37: 90-101. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/17560126
- Laufs H, Krakow K, Sterzer P, Eger E, Beyerle A, et al. Electroencephalographic Signatures of Attentional and Cognitive Default Modes in Spontaneous Brain Activity Fluctuations at Rest. Proc Natl Acad Sci U S A. 2003; 100: 11053-11058. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/12958209
- Fox M, Snyder AZ, Vincent JL, Corbetta M, Van Essen DC, et al. The Human Brain Is Intrinsically Organized into Dynamic, Anticorrelated Functional Networks. Proc Natl Acad Sci U S A. 2005; 102: 9673-9678. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15976020
- Greicius MD, Krasnow B, Reiss AL, Menon V. Functional Connectivity in the Resting Brain: A Network Analysis of the Default Mode Hypothesis. Proc Natl Acad Sci U S A. 2003; 100: 253-258. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/12506194
- Fransson P. Spontaneous low-frequency BOLD signal fluctuations: An fMRI investigation of the resting-state default mode of brain function hypothesis. Hum Brain Mapp. 2005; 26: 15-29. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15852468
- Vincent JL, Snyder AZ, Fox MD, Shannon BJ, Andrews JR. Coherent Spontaneous Activity Identifies a Hippocampal-Parietal Memory Network. J Neurophysio. 2006; 96: 3517-3531. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/16899645
- Rombouts SARB, Stam CJ, Kuijer JPA, Scheltens P, Barkhof F. Identifying confounds to increase specificity during a "no task condition". NeuroImage. 2003; 20: 1236-1245. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/14568492
- Horwitz B. The elusive concept of brain connectivity. NeuroImage. 2003; 19: 466-470.
- O'Kane G, Kensinger EA, Corkin S. Evidence for semantic learning in profound amnesia: An investigation with patient H.M. Hippocampus. 2004; 14; 417-425. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15224979
- Lehn H, Steffenach HA, van Strien NM, Veltman DJ, Witter MP. A specific role of the human hippocampus in recall of temporal sequences. The J Neurosci. 2009; 29: 3475-3484. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/19295153
- Merkow MB, Burke JF, Kahana MJ. The human hippocampus contributes to both the recollection and familiarity components of recognition memory. Proc Natl Acad Sci U S A. 2015; 112: 14378-14383. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26578784
- Harand C, Bertran F, La Joie R, Landeau B. The Hippocampus Remains Activated over the Long Term for the Retrieval of Truly Episodic Memories. PloS one. 2012; 7: e43495). PubMed: https://www.ncbi.nlm.nih.gov/pubmed/22937055
- Ezzati A, Katz MJ, Zammit AR, Lipton ML. Zimmerman ME, et al. Differential association of left and right hippocampal volumes with verbal episodic and spatial memory in older adults. Neuropsychologia. 2016; 93: 380-385. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/27542320
- Van Hoesen GW. The parahippocampal gyrus: New observations regarding its cortical connections in the monkey. Trends in Neurosciences. 1982; 5: 345-350.
- Bohbot VD, Allen JJB, Dagher A, Dumoulin SO, Evans AC, et al. Role of the parahippocampal cortex in memory for the configuration but not the identity of objects: converging evidence from patients with selective thermal lesions and fMRI. Frontiers in human neuroscience. 2015; 9: 431. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/26283949
- Kohler S, Danckert S, Gati JS, Menon RS. Novelty responses to relational and non-relational information in the hippocampus and the parahippocampal region: A comparison based on event-related fMRI. Hippocampus. 2005; 15: 763-774. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15999342
- WIttmann BC, Bunzeck N, Dolan RJ, Du E. Anticipation of novelty recruits reward system and hippocampus while promoting recollection. NeuroImage. 2007; 38: 194-202. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/17764976
- Howard LR, Kumaran D, Olafsdottir HF, Spiers HJ. Double Dissociation between Hippocampal and Parahippocampal Responses to Object-Background Context and Scene Novelty. J Neurosci. 2011; 31: 5253-5261. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/21471360
- Nielsen FA, Balslev D, Hansen LK. Mining the posterior cingulate: Segregation between memory and pain components. NeuroImage. 2005; 27: 520-532. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/15946864
- Sestieri C, Corbetta M, Romani GL, Shulman GL. Episodic Memory Retrieval, Parietal Cortex, and the Default Mode Network: Functional and Topographic Analyses. J Neurosci. 2011; 31: 4407-4420. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/21430142
- Raichle ME, MacLeod AM, Snyder AZ, Powers WJ, Gusnard DA, et al. A default mode of brain function. Proceedings of the National Academy of Sciences. 2001; 98: 676-682.
- Buckner RL, Andrews-Hanna JR, Schacter DL. The brain's default network. Annals of the New York Academy of Sciences. 1124; 1-38.
- Gusnard DA, Akbudak E, Shulman GL, Raichle ME. Medial prefrontal cortex and self-referential mental activity: relation to a default mode of brain function. Proc Natl Acad Sci U S A. 2001; 98: 4259-4264. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/11259662
- Addis DR, Wong AT, Schacter DL. Remembering the past and imagining the future: Common and distinct neural substrates during event construction and elaboration. Neuropsychologia. 2007; 45: 1363-1377. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/17126370
- Leech R, Kamourieh S, Beckmann CF, Sharp DJ. Fractionating the Default Mode Network: Distinct Contributions of the Ventral and Dorsal Posterior Cingulate Cortex to Cognitive Control. J Neurosci. 2011; 31: 3217-3224. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/21368033
- Mason MF, Norton MI, Van Horn JD, Wegner DM, Grafton ST, et al. Wandering Minds: The Default Network and Stimulus-Independent Thought. Science. 2007; 315: 393-395. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/17234951
Figures:
Figure 1
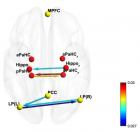
Figure 2
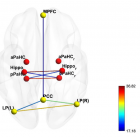
Figure 3
Figure 4
Similar Articles
-
Analysis of early Versus Delayed Carotid Surgery after Acute Ischemic StrokePEROU Sébastien*,DETANTE Olivier,SPEAR Rafaelle,PIRVU Augustin,ELIE Amandine,MAGNE Jean-Luc. Analysis of early Versus Delayed Carotid Surgery after Acute Ischemic Stroke. . 2017 doi: 10.29328/journal.jnnd.1001001; 1: 001-011
-
The “Calcium Paradox” Due To Ca2+/Camp Interaction: New Insights for the Neuroscience FieldLeandro Bueno Bergantin*, Afonso Caricati-Neto. The “Calcium Paradox” Due To Ca2+/Camp Interaction: New Insights for the Neuroscience Field. . 2017 doi: 10.29328/journal.jnnd.1001002; 1: 012-015
-
Protective functions of AEURA in Cell Based Model of Stroke and Alzheimer diseaseJigar Modi,Ahmed Altamimi,Ashleigh Morrell,Hongyuan Chou,Janet Menzie,Andrew Weiss,Michael L Marshall, Andrew Li,Howard Prentice*,Jang-Yen Wu*. Protective functions of AEURA in Cell Based Model of Stroke and Alzheimer disease. . 2017 doi: 10.29328/journal.jnnd.1001003; 1: 016-023
-
Direct Carotid Puncture for Flow Diverter Stent InsertionBhogal P*,Phillips TJ, Makalanda HLD. Direct Carotid Puncture for Flow Diverter Stent Insertion. . 2017 doi: 10.29328/journal.jnnd.1001004; 1: 024-028
-
Lateralized Cerebral Amyloid Angiopathy presenting with recurrent Lacunar Ischemic StrokeYi Li*, Ayman Al-Salaimeh,Elizabeth DeGrush,Majaz Moonis*. Lateralized Cerebral Amyloid Angiopathy presenting with recurrent Lacunar Ischemic Stroke. . 2017 doi: 10.29328/journal.jnnd.1001005; 1: 029-032
-
Experimental ‘hindbrain related’ syringomyelia: some mechanisms of spinal cord damageSergey N Larionov*,Sorokovikov VA,Rudakova AV. Experimental ‘hindbrain related’ syringomyelia: some mechanisms of spinal cord damage. . 2017 doi: 10.29328/journal.jnnd.1001006; 1: 033-038
-
Focal Ab-amyloid deposition precedes cerebral microbleeds and Superficial siderosis: a case reportParnesh Raniga,Patricia Desmond, Paul Yates,Olivier Salvado, Pierrick Bourgeat,Jurgen Fripp,Svetlana Pejoska, Michael Woodward,Colin L Masters,Christopher C Rowe,Victor L Villemagne*. Focal Ab-amyloid deposition precedes cerebral microbleeds and Superficial siderosis: a case report. . 2017 doi: 10.29328/journal.jnnd.1001007; 1: 039-044
-
Comorbidity of alcohol dependence with attention-deficit/hyperactivity disorder and the role of executive dysfunctionsCaterina Pistarini*,Gloria Tosi,Giovanni Vittadini,Ines Giorgi,Elena Fiabane,Paola Palladino. Comorbidity of alcohol dependence with attention-deficit/hyperactivity disorder and the role of executive dysfunctions . . 2018 doi: 10.29328/journal.jnnd.1001008; 2: 001-010
-
Spinal muscular atrophy counteracted by Agrin biological NT-1654Jes Paul*. Spinal muscular atrophy counteracted by Agrin biological NT-1654. . 2018 doi: 10.29328/journal.jnnd.1001009; 2: 011-013
-
Brain and immune system: KURU disease a toxicological process?Luisetto M*,Behzad Nili-Ahmadabadi,Ghulam Rasool Mashori,Ahmed Yesvi,Ram Kumar Sahu,Heba Nasser,Cabianca luca,Farhan Ahmad Khan. Brain and immune system: KURU disease a toxicological process?. . 2018 doi: 10.29328/journal.jnnd.1001010; 2: 014-027
Recently Viewed
-
Prediction of neonatal and maternal index based on development and population indicators: a global ecological studySedigheh Abdollahpour,Hamid Heidarian Miri,Talat Khadivzadeh*. Prediction of neonatal and maternal index based on development and population indicators: a global ecological study. Clin J Obstet Gynecol. 2021: doi: 10.29328/journal.cjog.1001096; 4: 101-105
-
A Genetic study in assisted reproduction and the risk of congenital anomaliesKaparelioti Chrysoula,Koniari Eleni*,Efthymiou Vasiliki,Loutradis Dimitrios,Chrousos George,Fryssira Eleni. A Genetic study in assisted reproduction and the risk of congenital anomalies. Clin J Obstet Gynecol. 2021: doi: 10.29328/journal.cjog.1001095; 4: 096-100
-
Leiomyosarcoma in pregnancy: Incidental finding during routine caesarean sectionToon Wen Tang*,Phoon Wai Leng Jessie. Leiomyosarcoma in pregnancy: Incidental finding during routine caesarean section. Clin J Obstet Gynecol. 2021: doi: 10.29328/journal.cjog.1001094; 4: 092-095
-
Adult Neurogenesis: A Review of Current Perspectives and Implications for Neuroscience ResearchAlex, Gideon S*,Olanrewaju Oluwaseun Oke,Joy Wilberforce Ekokojde,Tolulope Judah Gbayisomore,Martina C. Anene-Ogbe,Farounbi Glory,Joshua Ayodele Yusuf. Adult Neurogenesis: A Review of Current Perspectives and Implications for Neuroscience Research. J Neurosci Neurol Disord. 2024: doi: 10.29328/journal.jnnd.1001102; 8: 106-114
-
Late discover of a traumatic cardiac injury: Case reportBenlafqih C,Bouhdadi H*,Bakkali A,Rhissassi J,Sayah R,Laaroussi M. Late discover of a traumatic cardiac injury: Case report. J Cardiol Cardiovasc Med. 2019: doi: 10.29328/journal.jccm.1001048; 4: 100-102
Most Viewed
-
Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth EnhancersH Pérez-Aguilar*, M Lacruz-Asaro, F Arán-Ais. Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth Enhancers. J Plant Sci Phytopathol. 2023 doi: 10.29328/journal.jpsp.1001104; 7: 042-047
-
Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case PresentationJulian A Purrinos*, Ramzi Younis. Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case Presentation. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001099; 8: 075-077
-
Feasibility study of magnetic sensing for detecting single-neuron action potentialsDenis Tonini,Kai Wu,Renata Saha,Jian-Ping Wang*. Feasibility study of magnetic sensing for detecting single-neuron action potentials. Ann Biomed Sci Eng. 2022 doi: 10.29328/journal.abse.1001018; 6: 019-029
-
Pediatric Dysgerminoma: Unveiling a Rare Ovarian TumorFaten Limaiem*, Khalil Saffar, Ahmed Halouani. Pediatric Dysgerminoma: Unveiling a Rare Ovarian Tumor. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001087; 8: 010-013
-
Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative reviewKhashayar Maroufi*. Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative review. J Sports Med Ther. 2021 doi: 10.29328/journal.jsmt.1001051; 6: 001-007

HSPI: We're glad you're here. Please click "create a new Query" if you are a new visitor to our website and need further information from us.
If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."